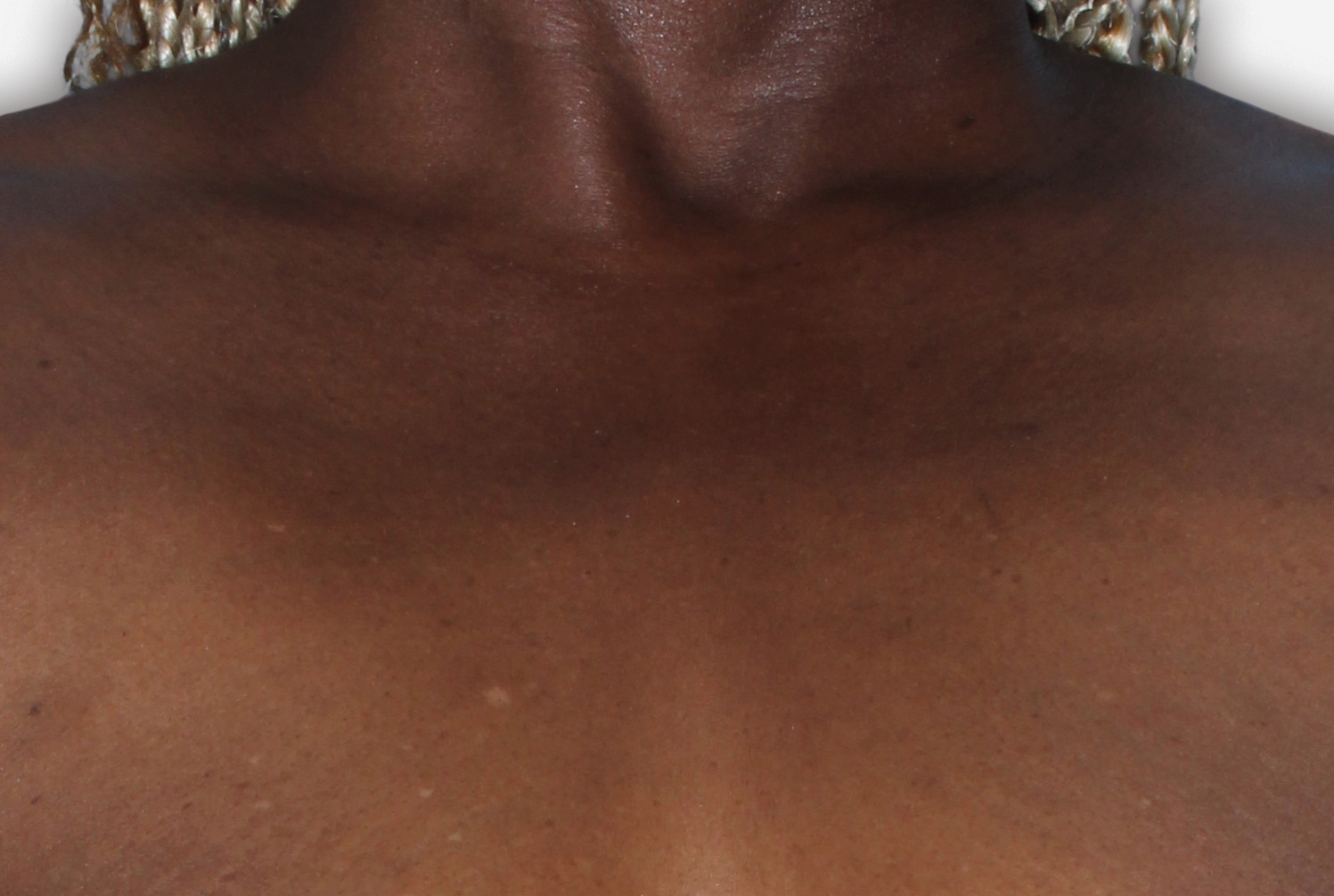
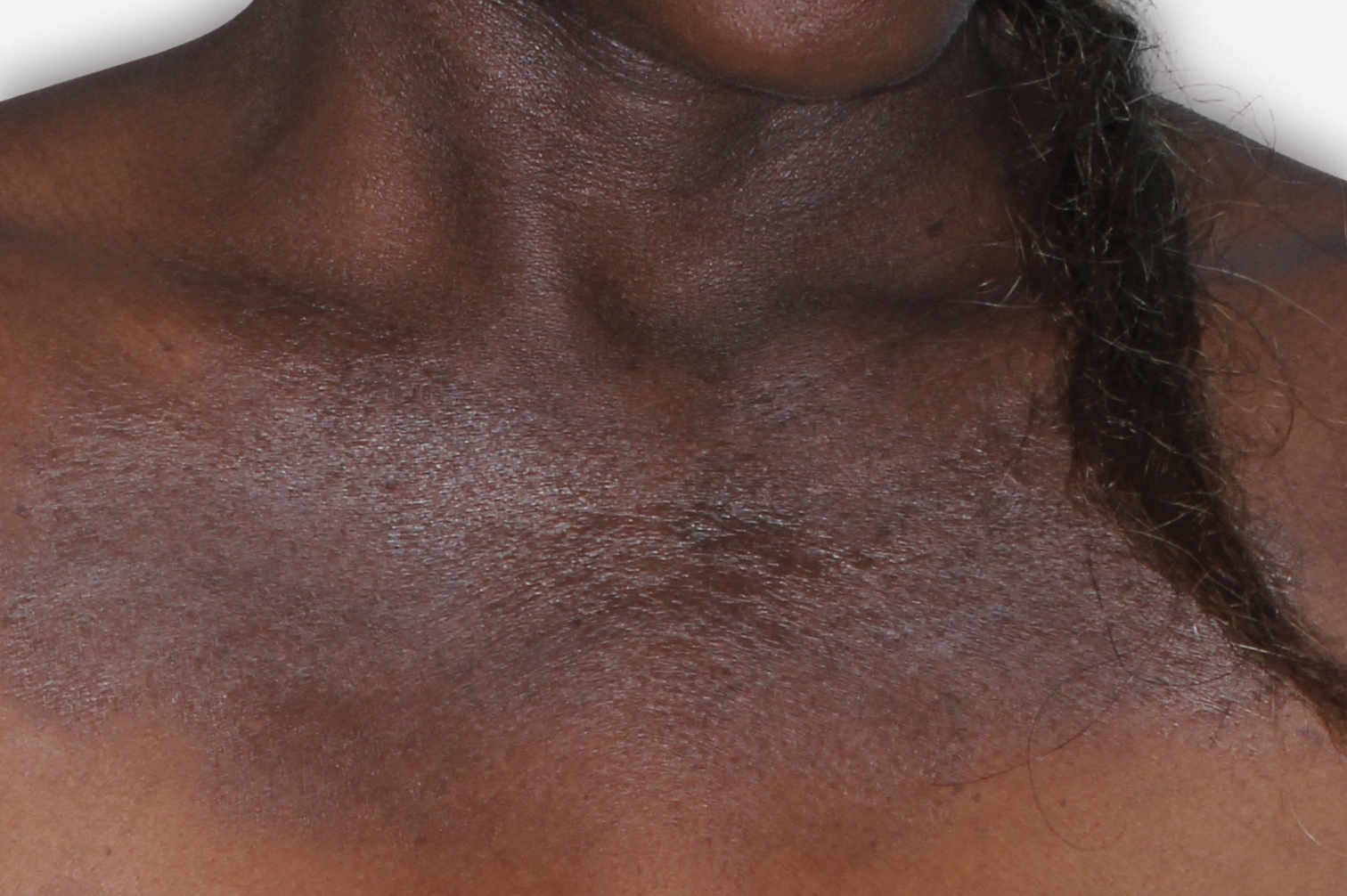
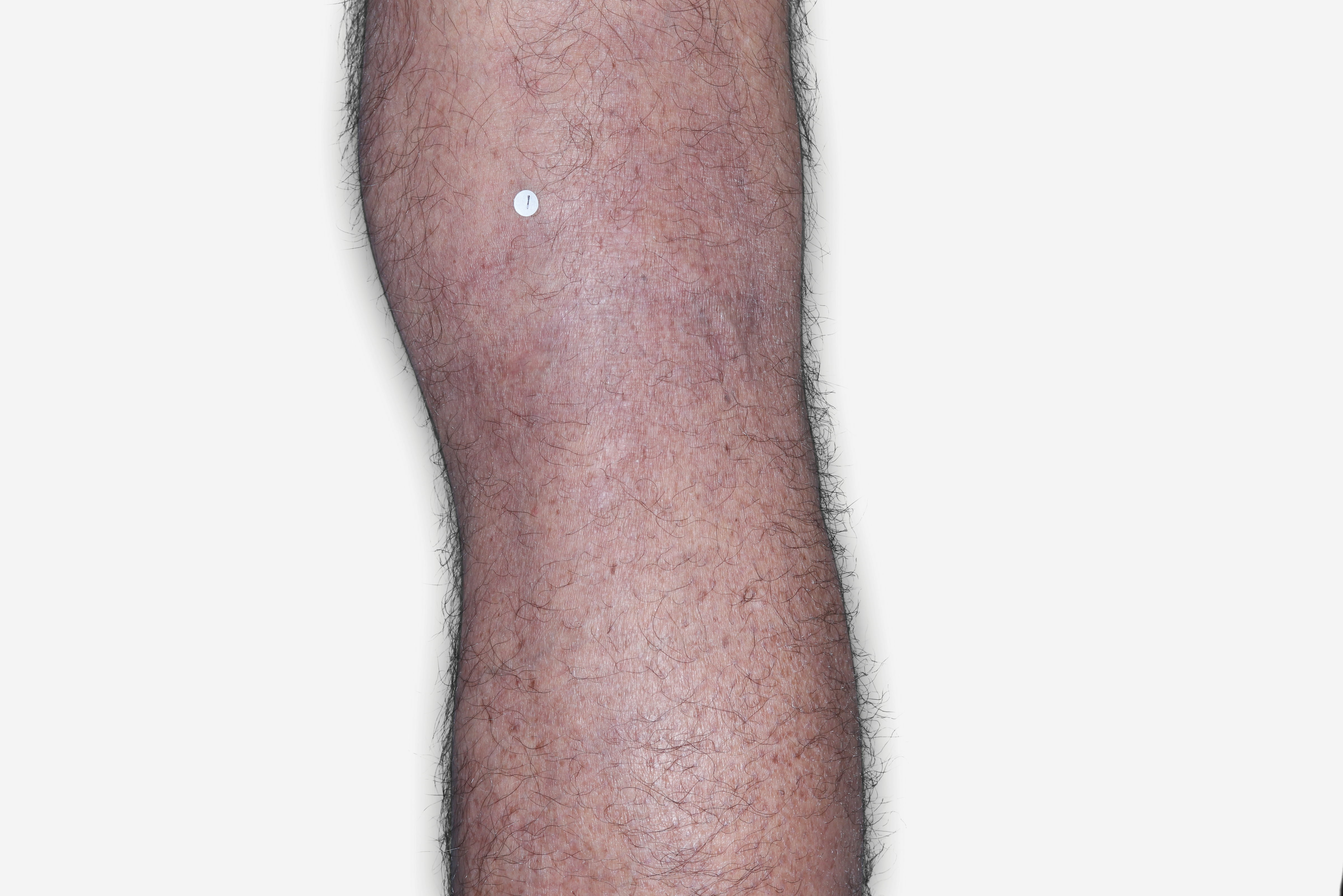
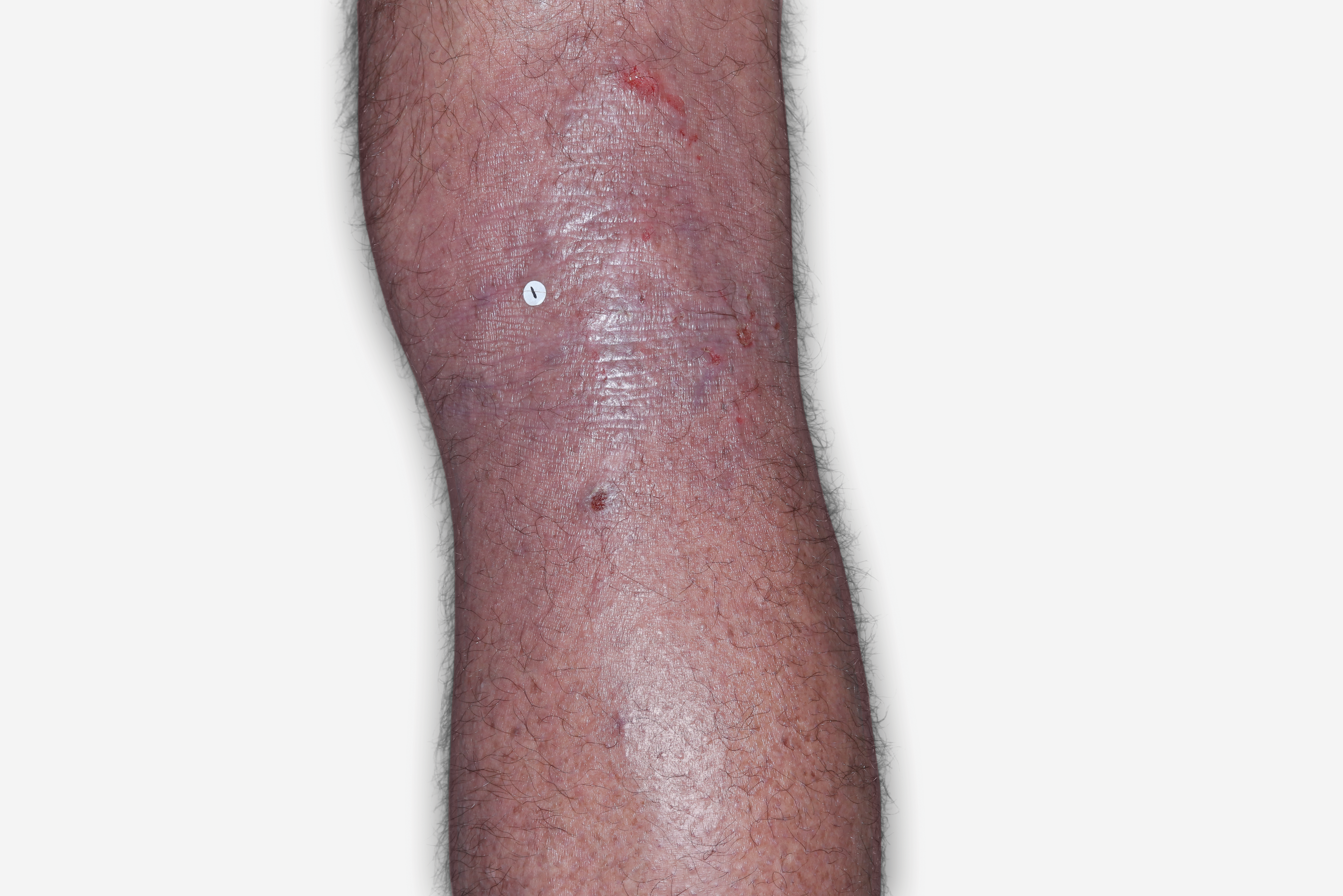
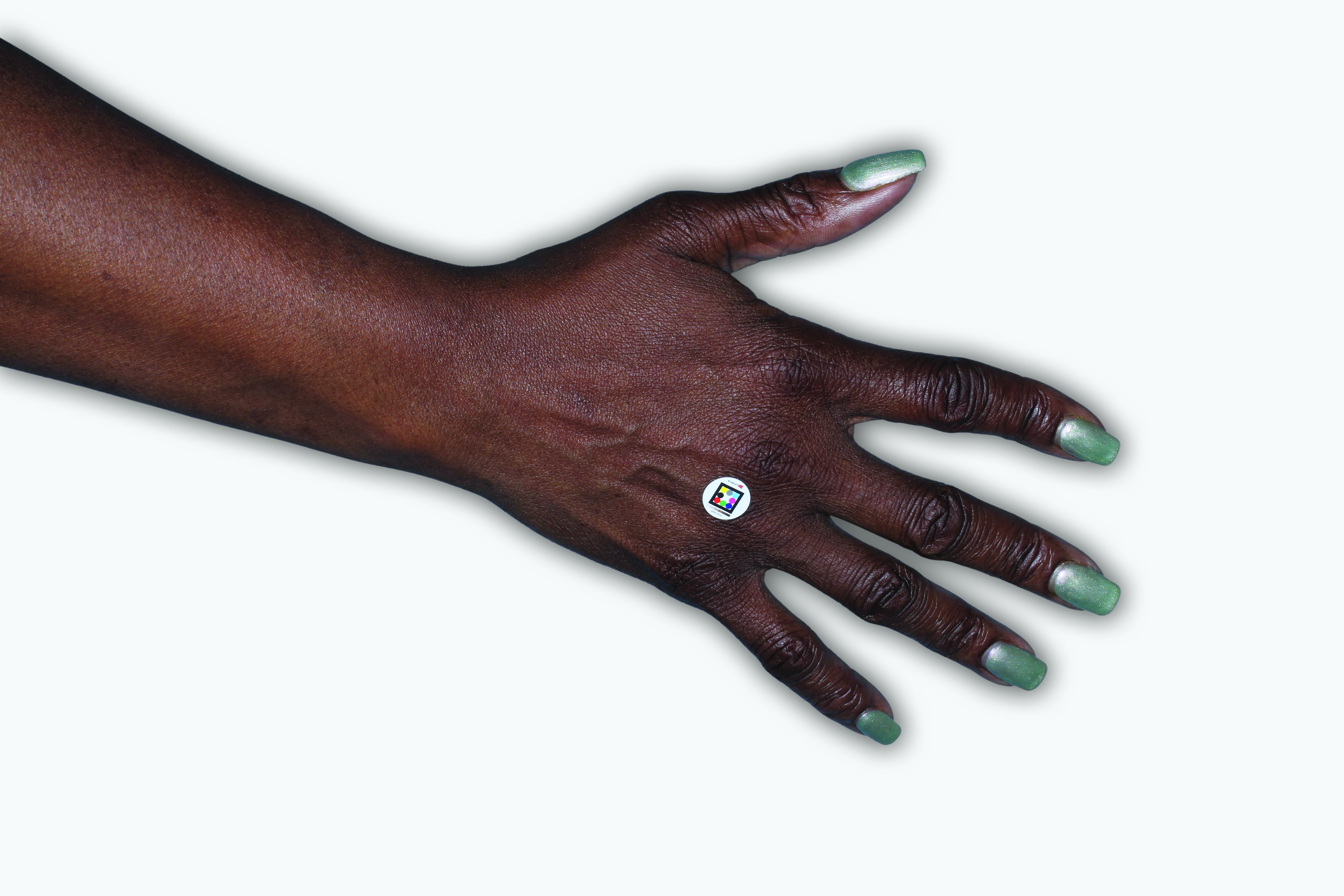
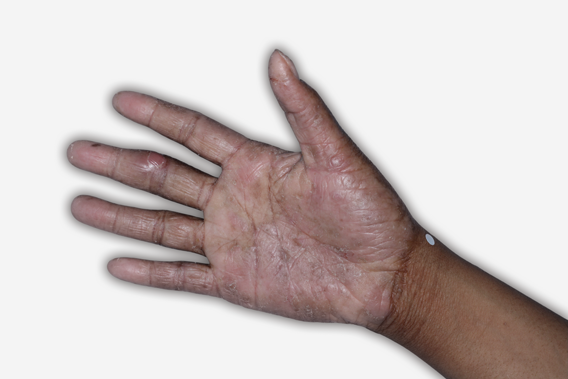
By enrolling in the EBGLYSS Savings Card Program (“Program”) and using the EBGLYSS Savings Card (“Card”), you attest that you meet the eligibility criteria, agree to, and will comply with the terms and conditions described below:
Card Eligibility:
- You have been prescribed EBGLYSS (lebrikizumab-lbkz) consistent with FDA-approved product labeling;
- You are enrolled in a commercial insurance plan;
- You are not enrolled in any state, federal, or government funded healthcare program, including, without limitation, Medicaid, Medicare, Medicare Part D, Medicare Advantage, Medigap, DoD, VA, TRICARE®/CHAMPUS, or any state prescription drug assistance program;
- You are a resident of the United States or Puerto Rico; and
- You are 18 years of age or older.
Card Terms and Conditions:
For patients with commercial insurance coverage for EBGLYSS: You must have commercial insurance that covers EBGLYSS and a prescription consistent with FDA-approved product labeling to pay as little as $5 for a 1-month prescription fill of EBGLYSS. Month is defined as 28 days. Card must be first used by no later than 12/31/2025. Card savings are subject to a maximum monthly savings of wholesale acquisition cost plus usual and customary pharmacy charges and a separate maximum annual savings of up to $9,450 per calendar year. Participation in the Program requires a valid patient HIPAA authorization upon enrollment into the Program. Subject to Lilly USA, LLC’s right to terminate, rescind, revoke, or amend Card eligibility criteria and/or Card terms and conditions which may occur at Lilly’s sole discretion, without notice, and for any reason, Card expires and savings end on 12/31/2027 or 24 months after you first use the Card, whichever comes first.
For patients with commercial insurance who do not have coverage for EBGLYSS: You must have commercial insurance that does not cover EBGLYSS and a prescription consistent with FDA-approved product labeling to pay as little as $25 for a 1-month supply of EBGLYSS. Month is defined as 28 days. Card must be first used by no later than 12/31/2025. Participation in the $25 Program requires submission of a prior authorization (PA) and a coverage denial outcome prior to first prescription fill. For patients who enrolled in the $25 Program on or before May 31, 2025, to remain eligible for continued enrollment in the $25 Program, a new PA must be submitted with a denial outcome received by August 1, 2025, and by each August 1st thereafter and as required by Lilly at its sole discretion. For patients who enrolled in the $25 Program on or after June 1, 2025, to remain eligible for continued enrollment in the $25 Program, a new PA must be submitted with a denial outcome received by August 1, 2026, and by each August 1st thereafter and as required by Lilly at its sole discretion. Card savings are subject to a maximum monthly savings and a separate maximum annual savings. Participation in the Program requires a valid patient HIPAA authorization to remain in the Program. Subject to Lilly USA, LLC’s right to terminate, rescind, revoke, or amend Card eligibility criteria and/or Card terms and conditions, which may occur at Lilly’s sole discretion, without notice, and for any reason, Card expires and savings end on 12/31/2027 or 24 months after you first use the Card, whichever comes first.
If you have an insurance plan that is participating in an alternate funding program (AFP) that requires you to apply to the EBGLYSS Savings Card Program or otherwise pursue specialty drug prescription coverage through an alternate funding vendor as a condition of, requirement for, or prerequisite to coverage of EBGLYSS, you are not eligible for and are prohibited from using the EBGLYSS Savings Card Program. AFPs include programs where coverage, reimbursement, or patient out of pocket costs for a product in some way vary based on the availability of a manufacturer co-pay program. AFPs may modify, delay, deny, restrict, or withhold insurance benefits or coverage from patients, or exclude Lilly products from coverage contingent upon a member’s use of EBGLYSS Savings Card Program. You agree to inform EBGLYSS Savings Card Program if you are or become a member of such an alternative funding program. You are responsible for any applicable taxes, fees, and any amount that exceeds the monthly or annual maximum Card savings. Monthly and annual maximum savings are set at Lilly’s sole and absolute discretion and may be changed with or without notice at any time for any reason. At its sole discretion and with or without notice, Lilly may reduce, eliminate, or otherwise modify the Card savings for any reason, including but not limited to if your commercial drug insurance plan imposes additional requirements which limits or prevents you from receiving coverage for EBGLYSS, only allows partial coverage for EBGLYSS, removes coverage for EBGLYSS and requires you to utilize the Card, does not provide a material level of financial assistance for the cost of EBGLYSS, or does not apply Card payments to satisfy your co-payment, deductible, or coinsurance for EBGLYSS. Card savings are not valid for: Massachusetts residents if an AB-rated generic equivalent is available; California residents if an FDA-approved therapeutic equivalent is available. You must meet the Card eligibility criteria, terms and conditions every time you use the Card. Card activation is required. You may not seek reimbursement from your health insurance, any third party, or any health savings, flexible spending, or other healthcare reimbursement accounts, for any amount of the savings received through the Card. By utilizing the Card, you agree that if you are required to do so under the terms of your insurance coverage for this prescription or are otherwise required to do so by law, you will notify your Insurance Carrier of your redemption of the Card. Card savings cannot be combined or utilized with any other program, discount, discount card, cash discount card, coupon, incentive, or similar offer involving EBGLYSS. You agree that this Card savings is intended solely for the benefit of you, the patient, and that the Card benefits are nontransferable. It is prohibited for any person to sell, purchase, or trade; or to offer to sell, purchase, or trade, or to counterfeit the Card. The Card is not insurance. Lilly has the sole right to interpret and apply Card eligibility criteria, and terms and conditions. Card eligibility, and terms and conditions may be terminated, rescinded, revoked, or amended by Lilly at any time without notice and for any reason. Lilly’s sole discretion to terminate, rescind, revoke, or amend Card eligibility criteria and/or Card terms and conditions includes the right to terminate any individual Card if Lilly determines, in its sole discretion, that a patient does not satisfy the Card’s eligibility criteria or is using or has attempted to use the Card inconsistently with these Terms and Conditions. Eligibility criteria, and terms and conditions for the EBGLYSS Savings Card Program may change from time to time; the most current version can be found at https://www.EBGLYSS.lilly.com/savings-support#termsandconditions. You may be required to obtain a new Card, including if any Card terms and conditions have been terminated, rescinded, revoked, or amended by Lilly. Card void where prohibited by law. Subject to Lilly’s right to terminate, rescind, revoke or amend Card eligibility criteria and/or Card terms and conditions, which may occur at Lilly’s sole discretion, without notice, and for any reason, the Card expires and savings end on 12/31/2027 or 24 months after you first use the Card, whichever comes first.